Antibody
Conjugation
Start your project
Any payload, any format
Antibody Conjugate fitting your science
We specialize in tailored, high-performance immunoconjugate production, including ADCs, DACs, and AOCs. Technology-agnostic by design, we work across all conjugation formats and platforms. From feasibility to preclinical supply, we support your program with flexible formats, multiple conjugation technologies, and comprehensive analytical packages.
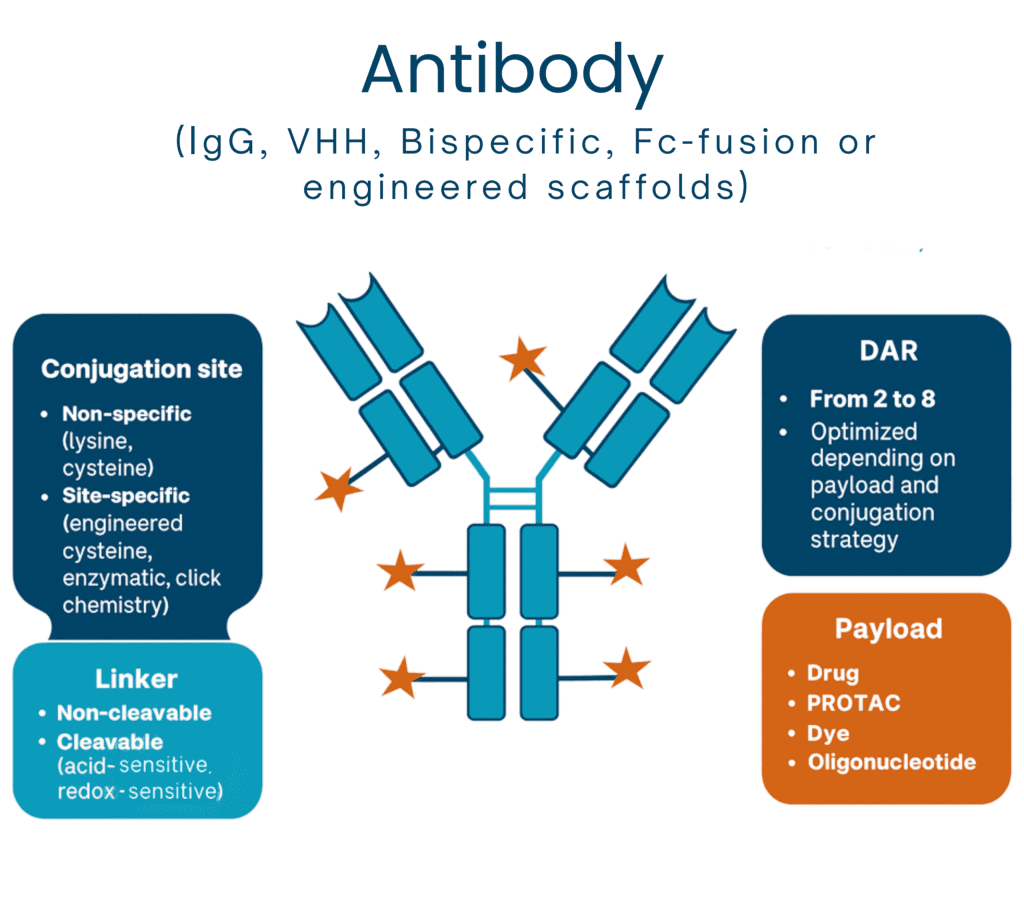
Custom reliable Antibody Conjugate
Any antibody format and multiple conjugation strategies. We deliver high-quality, QC-controlled antibody conjugates
Derisk your project
Expert guidance to accelerate antibody conjugate development from concept to scale
Full Service Approach
Achieve program success through our end-to-end integrated platform
Tailored Antibody Conjugates without Compromise

More than 220 Ab conjugates produced
Proven track record across diverse antibody formats, payloads, and conjugation chemistries.

From milligram to gram-scale
Supporting discovery, preclinical, and scale-up phases with flexible production capabilities.
Custom fully Integrated Project Solutions
We adapt to your needs and built antibody conjugates to your specifications.

Quality assurance
ISO 9001 certification guarantees the quality of our services.
- Concentration determination
- DAR determination
- Residual toxin estimation
- Monomeric purity
- Hydrophobicity profiling
- Endotoxin level assessment
Our Expertise Across Multiple Conjugation Strategies
We developed a wide range of robust strategies to conjugates IgGs, scFvs, bispecifics, or VHHs to linkers and payloads, including site-specific, cysteine (DAR 4–8), enzymatic (BTG), or click chemistry.
Explore the table below for all available options.
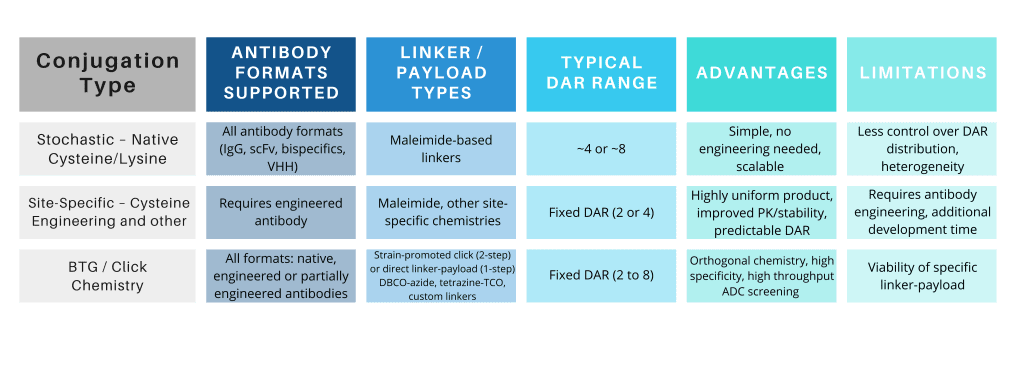
Payload compatibility

ADC library
Our ADC Library program accelerates development from candidate antibodies, enabling early efficacy evaluation and informed decision-making.
The program includes :
Purified ADC samples with defined DAR, QC reports, functional assay data, and a technical dossier summarizing conjugation and analytical results.
