Protein
Bioproduction
Start your project
Antibodies Made to Measure
Any Format, Any Scale
Our company is specialized in antibody production, offering custom formats at various scales while consistently meeting the required specifications.
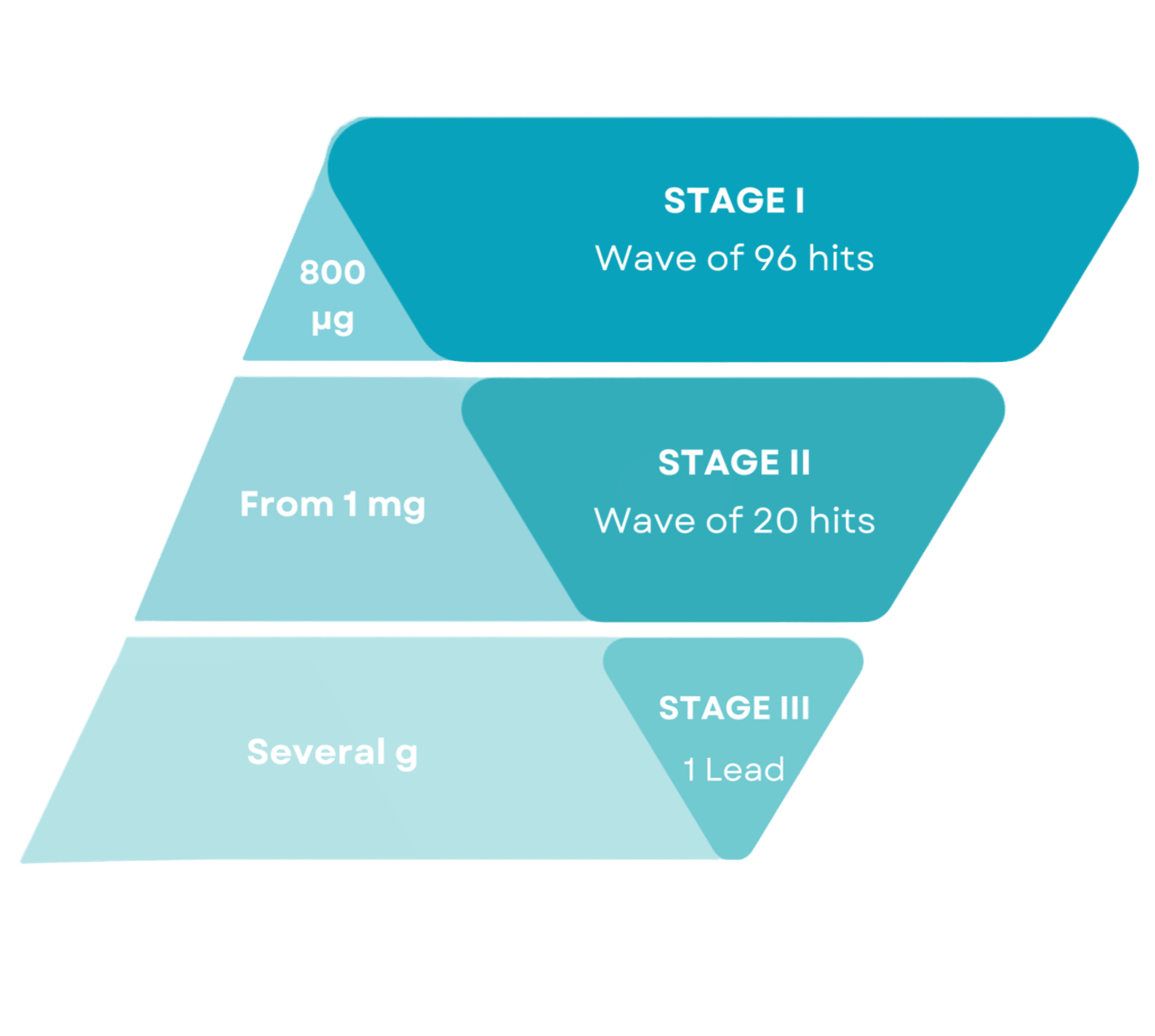
Stage 1
High Throughout production at Microscale Hit discovery & screening
Stage 2
Small scale production
Lead selection
Stage 3
Medium & Large-scale production
Lead validation
Custom reliable antibodies
We bring you exceptional results.
Fuel Research
We offer precision antibody solutions.
Achieve tailor-made antibody success
We offer an integrated development process.

European Production
To support researchers, biotech, startups and pharma labs with tailored solutions.

From micrograms to grams
We deliver the quantity you need for your projects.
Custom Quotes or Integrated Project Solutions
Our comprehensive quality checks guarantee that every batch matches your exact needs.

Quality assurance
ISO 9001 certification guarantees the quality of our services.
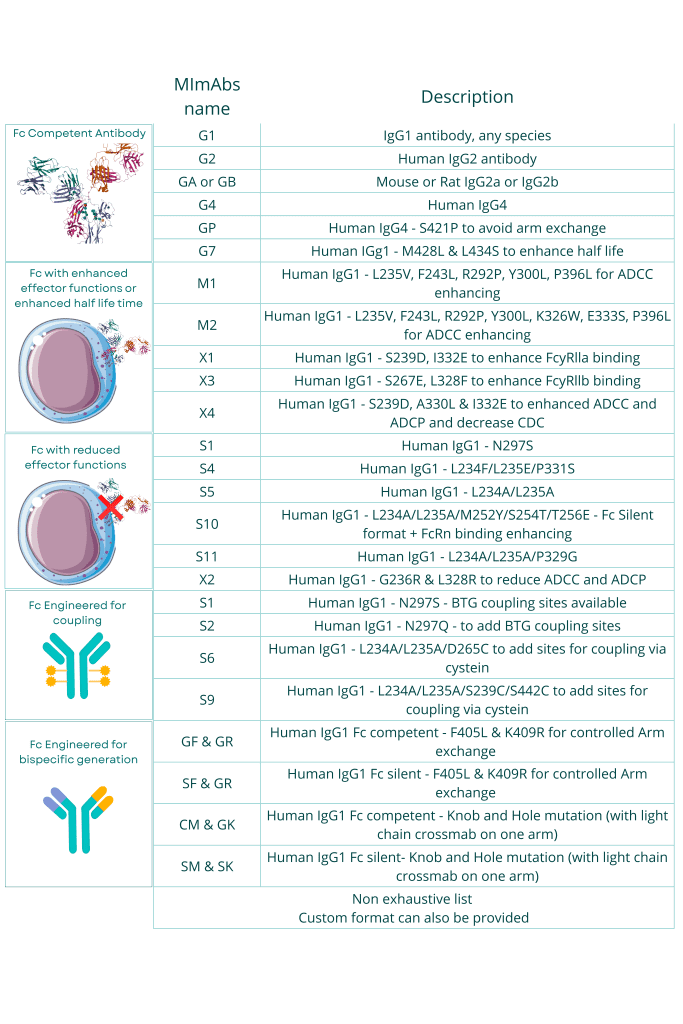
Classical of custom-made antibody formats can be produced based on client-specific requirements, such as monospecific, multispecific antibodies and antibody fragments (e.g., Fab, scFv, VHH).
High-throughout antibody production
Express up to 96 antibodies in HEK293 systems, with custom formats, for functional screening such as EC50 determination, binding, and/or affinity assays.
HEK293 Expression System



- Concentration determination
- Nucleic acid contamination evaluation

- Productivity
- Fragmentation and purity by SDS-PAGE
- Aggregation and monomeric purity by SEC-UPLC
- Endotoxin level
- Antibody mass by LC-MS
From small scale to large scale production
Accelerate your antibody development with flexible production, tailored to your needs. Express antibodies in HEK293 or CHO within weeks, in the desired format, from milligram to gram-scale yields. You’ll have the material you need for everything from in vitro assays to ADC generation and in vivo studies.
HEK or CHO Expression System



- Concentration determination
- Nucleic acid contamination evaluation
- Fragmentation and purity by SDS-PAGE
- Agregation and monomeric purity by SEC-UPLC
- Endotoxin level
- Antibody mass verification by LC-MS
Our custom formats
From antigen design to final production
We offer end-to-end support with scientific rigor and flexibility. Whether for microscale R&D or preclinical studies, you can count on a reliable, responsive partner.
See the serviceTrusted by scientists
As specialists in custom monoclonal antibody production, we support researchers, biotechs, startups and pharmas with tailored solutions.
Driven by quality. Built for innovation.
